Eating clean and sleeping well still isn’t enough. A protein your body makes more of every year is quietly doing the real damage.
You follow a healthy lifestyle. You sleep reasonably well. You eat a balanced diet. You exercise regularly. And yet there’s a kind of slow decline in physical and mental function that no amount of effort seems to stop.
Your joints ache a little longer. Recovery takes more out of you. Your face looks incrementally older every month, almost like time has quietly sped up.
Most people assume this is just how aging works. You get older. Things break down. Accept it. But that assumption has a problem: two people of the same chronological age can have biological systems that differ by fifteen years or more. One ages slowly. The other is already decades ahead in cellular decline. The difference runs deeper than lifestyle. It’s something happening at a molecular level that most mainstream health writing never touches.
And scientists may have found the protein responsible for flipping that switch.
This article covers a lot of molecular ground. There is a plain-language reference for every scientific term used towards the end of the article, which can easily be accessed from the table of contents.
The Inflammaging Paradox
Most aging research focuses on what you’re doing wrong. It largely ignores what the body is already doing to itself. Even if you follow every guideline, a process runs in the background that steadily accumulates damage regardless. The mechanism has a name: inflammaging. It was defined in a 2018 Nature Reviews Endocrinology paper as a chronic low-grade inflammation that quietly drives all major age-related diseases. It’s not the same as the acute inflammation experienced when you sprain an ankle. There’s no redness, no obvious pain. It just runs continuously, like a furnace left on low heat, slowly degrading everything around it.
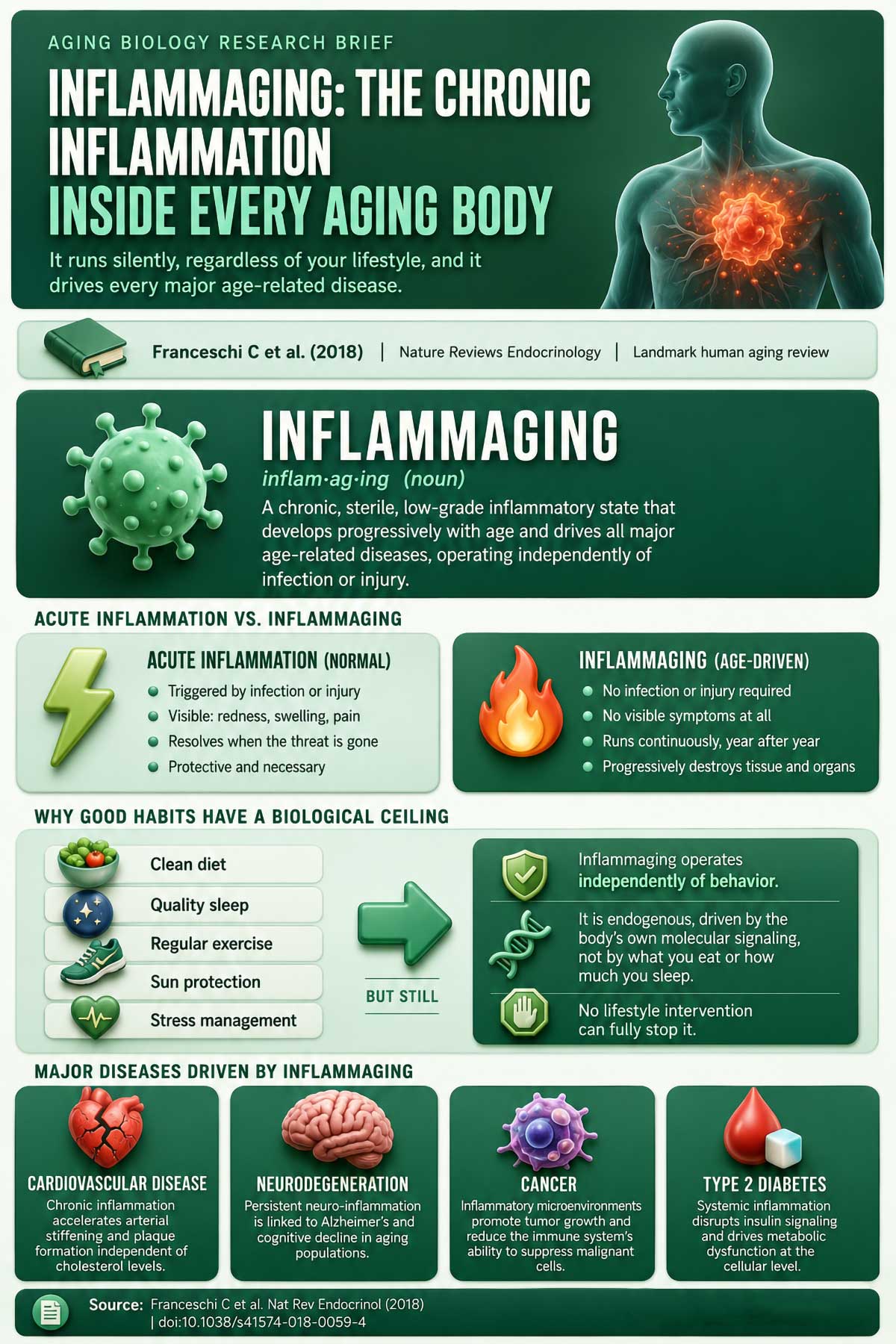
This is why the standard list of aging interventions has a biological ceiling. Sunscreen addresses UV damage. Better sleep reduces cortisol. A clean diet lowers glycemic stress. All of it helps. But none of it touches the internal inflammatory state that your body progressively deepens on its own, independent of your habits.
The urgency here goes beyond skin. Research published in Nature Medicine found that pro-inflammatory cytokines are the common denominator in about half of all deaths worldwide, spanning cardiovascular disease, cancer, and diabetes. Accelerated biological aging shows up long before any wrinkle does. Its real cost is measured in those same diseases, the ones that kill people early.
But to understand what’s actually driving that inflammation as you age, you need to look at one specific protein that most people have never heard of.
The Discovery of the Aging Switch
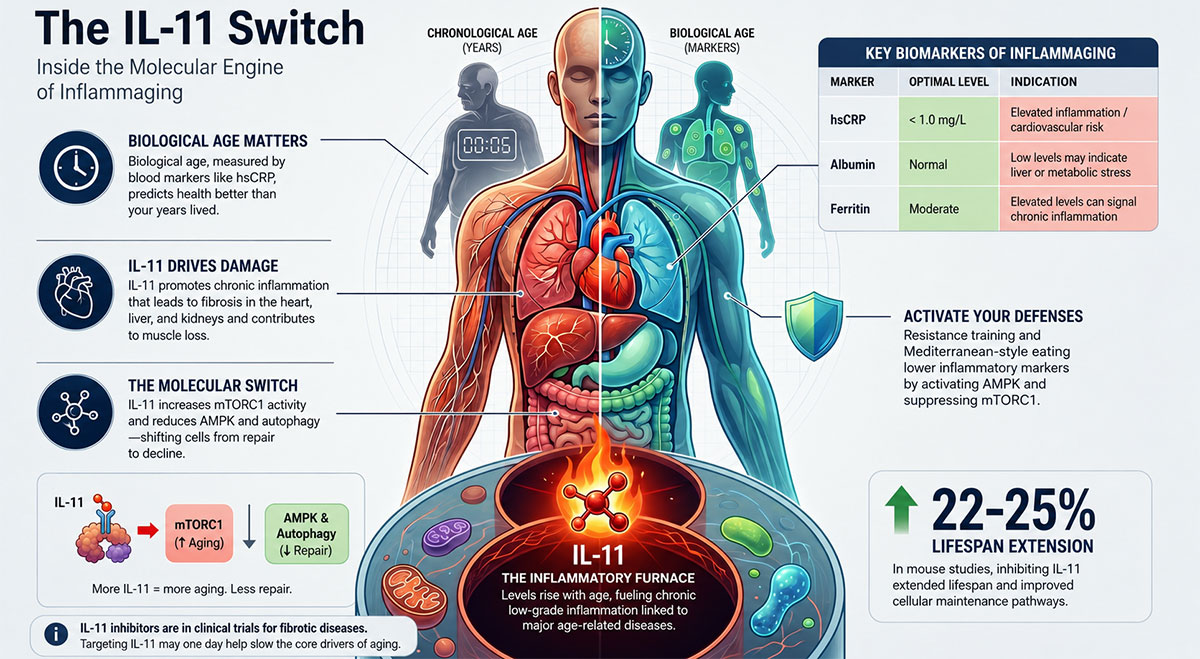
The protein is called IL-11. It belongs to a family of cytokines, the signaling molecules that tell cells what to do. For decades, it sat in the background of aging research, overshadowed by more well-known targets like IL-6. That changed dramatically with a 2024 Nature study that produced results almost nobody in mainstream science was expecting.
The researchers inhibited IL-11 in mice at approximately the equivalent of 55 human years, the age window when biological decline typically accelerates. The effect on lifespan was striking: median lifespan extended by 22.4% in males and 25% in females.

Blocking IL-11 suppressed mTORC1, a pathway that drives cellular aging. It also boosted AMPK and autophagy, the body’s internal repair and recycling systems. Those aren’t minor adjustments. They’re the central regulatory mechanisms that determine whether a cell repairs itself or deteriorates into dysfunction.
This connects directly to the broader scientific framework of aging. Chronic inflammation is now listed as a distinct driver of aging alongside the other eleven identified mechanisms. IL-11 inhibition, according to the evidence, addresses at least four of those twelve hallmarks simultaneously. That’s a degree of reach that no lifestyle intervention comes close to matching.
Why This Specific Protein Destroys the Body Over Time
Most of us think of protein as the ultimate “good guy” in biology. It is the fuel for our muscles and the foundation of a strong immune system. That is why it is so jarring to learn that one specific protein is actually a villain when it comes to aging. IL-11 is a complete outlier. It ignores the standard rules of how proteins are supposed to behave. Instead of helping your body repair itself, it acts as a signal for your organs to start stiffening and your muscles to waste away. It is essentially a biological glitch that makes getting older much harder than it needs to be.
The real issue is that IL-11 levels start to increase as we age, which effectively signals the body to stop maintaining itself. This increase is what triggers sarcopenia, that steady loss of muscle and strength that makes staying active so much harder. It is basically a biological trap. The more of this protein you have in your system, the faster your metabolism and muscle mass decline.
Because the body does not have a built-in way to turn this signal off, it becomes a cycle of decay that just keeps feeding into itself. It is one of those frustrating internal glitches that explains why “getting old” feels like such an aggressive process.
The organ-level damage is even more concrete. A Nature study identified IL-11 as the dominant driver of fibrosis in human heart tissue, displacing previous assumptions that IL-6 or TGF-beta were the primary culprits.
Fibrosis is scar tissue forming inside organs as they age. The heart becomes stiffer. The liver loses function. The kidneys accumulate damage. The decay is real and structural, and IL-11 appears to be the signal that tells your organs to replace functional tissue with rigid, non-functional scarring.
The process begins well before any outward signs appear. By the time external markers of aging become visible, fibrosis in the major organs may already be years underway.
What Organ Fibrosis Actually Feels Like
Most people associate aging with how they look. But the internal progression of fibrosis has a distinct physical signature, and many people over 55 are already living with it.
Cardiac fibrosis makes the heart muscle less pliable. The result shows up as reduced ability to exercise, shortness of breath on effort that wasn’t there five years ago, and a recovery window after physical exertion that keeps getting longer. It’s easy to chalk this up to being out of shape. It’s often something more structural.
Liver fibrosis is even quieter. The liver has an enormous reserve capacity, so functional decline often produces no symptoms until a significant proportion of the organ is affected. Fatigue that sleep doesn’t fix is one of the few early signals. Slower detoxification, heightened sensitivity to alcohol, and difficulty maintaining stable energy across the day are other symptoms. These are vague enough that most people attribute them to stress or poor sleep rather than progressive organ change.
Kidney fibrosis follows a similar pattern. Subtle fluid retention, increased blood pressure that appears without an obvious cause, and a gradual decline in how efficiently the body clears metabolic waste are all early indicators. None of them announce themselves as organ damage. They present as things people accept as part of getting older. The science on IL-11 suggests they may be something more specific than that.
What You Can Do About Inflammaging Right Now
No lifestyle intervention stops inflammaging entirely. That ceiling is real. But the gap between doing nothing and doing the right things is significant, and several interventions have solid evidence for lowering the inflammatory markers most closely tied to biological aging.
Diet is the most well-supported tool. A review cited in a 2024 Aging Clinical and Experimental Research paper examined 46 studies and found that Mediterranean-style eating was steadily linked to lower inflammatory markers, including CRP and IL-6.
The PREDIMED trial went further. After three years, both Mediterranean diet groups showed clear reductions in IL-1β, IL-6, IL-8, and TNF-α compared with the control group. These are the same cytokine families that drive the inflammaging process. The food pattern itself isn’t complex: high intake of vegetables, legumes, oily fish, olive oil, and whole grains, combined with low intake of ultra-processed foods, refined sugars, and seed oils high in omega-6 fats.
Exercise works through the same cellular pathways that IL-11 blocking targets. Research in Frontiers in Immunology (2026) showed that resistance training in aging muscle dials down mTORC1 overactivation and raises AMPK activity. Those are the same two shifts seen in the Widjaja lifespan study.
It also triggers autophagy, the cell cleanup process that slows with age. In effect, regular resistance training is partly doing what IL-11 blocking does at the molecular level. It won’t close the gap entirely, but it engages the same repair systems.
Caloric restriction and time-restricted eating also have evidence behind them for AMPK activation and mTORC1 suppression, though how much they help in practice varies. The simpler version, eating within a set daily window and avoiding late-night meals, has shown real effects on inflammation markers in several human trials, without the difficulty of severe calorie cutting.
The evidence is more mixed on the supplement side. Quercetin and resveratrol have shown AMPK-activating effects in lab models and some human studies, but results in real-world aging outcomes aren’t yet proven. Omega-3 fats have stronger human data for lowering CRP and TNF-alpha. They’re the most defensible supplement choice from an inflammaging standpoint. Fish oil at doses used in clinical trials (2 to 4 grams of combined EPA and DHA daily) steadily reduces circulating inflammation markers in adults over 50.
Beyond Looking Old: The Reality of Accelerated Aging
Most people who search for what accelerates aging are thinking about their face. Fine lines. Sagging skin. Tired eyes. These are real, and they matter to people. But they’re the visible surface of a process that’s simultaneously dismantling the cardiovascular system, the liver, the kidneys, and the metabolic machinery that keeps everything else running.
This reframe matters because it changes what aging faster actually means. Looking older than your age is one signal. A more serious one is whether your biological clock is running ahead of your chronological one. That clock is measured by inflammation markers, cellular repair capacity, and organ function.
According to research, biological age driven by inflammation markers is a more accurate predictor of death than the number of years you’ve been alive. Chronological age is a count. Biological age is the actual state of the machine, and the gap between those two numbers is where the real risk lives.
Can You Actually Measure Your Biological Age?
The short answer is yes, and you may already have much of the data you need. The PhenoAge clock developed by Levine et al. is calculated from nine standard blood markers: albumin, creatinine, glucose, CRP, lymphocyte percentage, mean cell volume, red cell distribution width, alkaline phosphatase, and white blood cell count.
A standard metabolic panel, a complete blood count with differential, and a standalone CRP test provide all nine. If you’ve had thorough bloodwork recently, you may be able to calculate your PhenoAge now using publicly available calculators.
For those wanting a more detailed picture, commercial biological age testing has become much more accessible. InsideTracker analyses blood across 17 markers in men and 13 in women, using an algorithm to estimate biological age and flag which values are driving it higher.
TruDiagnostic does not bother with standard blood chemistry. It goes straight for your DNA methylation patterns instead. This gives you a breakdown for 11 different organs, so you can see if your heart is actually aging faster than your brain. The real reason people use this test is for the DunedinPACE metric. Most biological age tests are like an odometer showing your total miles, but this one is a speedometer that tells you how fast you are aging at this exact moment. It is definitely an investment, costing anywhere from $200 to $500, and they ship just about anywhere.
If you’re reading standard bloodwork for signs of inflammaging, here’s what to focus on. High-sensitivity CRP (hsCRP) below 1.0 mg/L is optimal. Above 3.0 mg/L signals elevated chronic inflammation.
Ferritin, while mainly an iron marker, also rises with chronic low-grade inflammation and tends to creep up with age. White blood cell count at the high end of normal (above 7.0 x10^9/L) has been linked to biological age acceleration in several large studies.
None of these markers is conclusive on its own, but together they give a useful snapshot of your inflammatory load. Worth raising with a doctor if any are elevated across more than one test.
Where the Science Is Heading
IL-11 inhibitors aren’t yet available as a treatment for aging. The Widjaja study used mice, and human trials would need to establish safety and efficacy at scale. That is a long road. But the road is shorter than most people realise.
Multiple IL-11-targeting antibodies are in active clinical trials right now. Boehringer Ingelheim launched a Phase IIa trial of its anti-IL-11 antibody BI 765423 in early 2026, testing its ability to halt fibrosis and restore lung function in patients with idiopathic pulmonary fibrosis.
Mabwell’s 9MW3811 has entered Phase 2 trials for pathological scarring and has received regulatory clearance in the US, China, and Australia. Lassen Therapeutics is testing LASN01, an anti-IL-11 receptor antibody, in patients with IPF and progressive fibrosing lung disease, with trial results expected in 2025 to 2026.
None of these trials targets aging directly. But fibrosis is the mechanism by which IL-11 wears organs down with age. Progress in one area feeds directly into the other.
What the research does offer right now, before any drug arrives, is a precise target. For decades, anti-aging science worked around the edges: reducing risk factors, improving sleep, cutting oxidative stress. All of it is useful. None of it is able to touch the core mechanism.
Finding IL-11 as the primary driver of the fibrotic, inflammatory cascade gives researchers a clear molecular address to work from. Everyone else gets a clearer picture of what the body is actually doing between birthdays.
The whole conversation about what makes you age faster has been aimed at habits and surfaces. The science on IL-11 suggests the real action has been one level below that all along, in a signaling loop the body runs quietly on its own. No permission asked. No symptoms given. And for which better sleep and more vegetables help, but not quite enough.
Glossary of Terms
This article covers a lot of molecular ground. Here’s a plain-language reference for every scientific term used.
AMPK (AMP-activated protein kinase)
AMPK is basically the master power switch for your cells. When you are low on energy, because you are fasting or working out, AMPK tells your body to stop trying to grow and start repairing what is already there. It is the biological signal for internal cleanup. The trouble is that as we age or eat too much, that switch gets stuck in the off position. Instead of fixing itself, the body stays in a constant state of growth, and that is how the cellular junk starts to pile up.
Albumin
The most abundant protein in blood plasma. Made by the liver, it carries hormones, enzymes, and other substances through the bloodstream. Low albumin is a marker of poor liver function and is one of the nine inputs used to calculate PhenoAge biological age.
Autophagy
The body’s cellular housekeeping process. Cells break down and recycle damaged or dysfunctional components to make way for new, healthy ones. Autophagy slows with age, and debris starts to accumulate. IL-11 suppresses it. Exercise and fasting activate it.
Biological age
A measure of how old your body is at the cellular and organ level, as distinct from how many years you’ve been alive. Two people who are both 58 chronologically can have biological ages that differ by a decade or more. Inflammation markers are among the strongest predictors of biological age.
Cardiac fibrosis
The replacement of flexible heart muscle tissue with stiff scar tissue. It makes the heart less efficient at pumping blood and less able to recover from physical exertion. IL-11 is the primary molecular driver of this process in aging adults.
Chronological age
The number of years you’ve been alive. A simple count that tells you how long you’ve existed, but nothing about the state of your cells, organs, or immune system.
CRP (C-reactive protein)
CRP is essentially an internal alarm bell that your liver rings whenever it detects inflammation. It is a common blood marker, but for most people, the standard test is too blunt. You really want hsCRP. It is much more sensitive, so it can pick up the quiet, low-grade inflammation that ages your organs over time. Think of it like a smoke detector that is sensitive enough to catch a smoldering wire before the whole house catches fire. If you are just looking at regular CRP, you might miss the very thing that is actually wearing your body down.
Creatinine
A waste product filtered from the blood by the kidneys. Elevated creatinine signals that the kidneys aren’t clearing waste efficiently. It’s one of the nine markers used in the PhenoAge biological age calculation.
Cytokine
A small signaling protein released by immune cells to communicate with other cells. Cytokines can switch inflammation on or off. Pro-inflammatory cytokines, including IL-11, IL-6, and TNF-alpha, become chronically elevated with age and drive organ damage over time.
DNA methylation
A chemical process where small tags are added to DNA at specific sites, affecting which genes are switched on or off. Methylation patterns change with age in predictable ways. That consistency makes them a reliable biological clock. Tests like TruDiagnostic read these patterns to estimate biological age.
DunedinPACE
A biological aging metric developed at Duke University. Rather than estimating how old you are, it measures how fast you’re currently aging, expressed as a rate. A score above 1.0 means you’re aging faster than average for your chronological age. It’s calculated from DNA methylation data.
EPA and DHA
Two omega-3 fatty acids found primarily in oily fish and fish oil supplements. Both have solid evidence for lowering circulating inflammation markers, including CRP and TNF-alpha. Clinical trials typically use combined doses of 2 to 4 grams per day to produce measurable anti-inflammatory effects.
Ferritin
A protein that stores iron inside cells. Blood ferritin levels reflect both iron stores and general inflammation. Ferritin tends to rise with age and chronic low-grade inflammation. That makes it a useful secondary marker of inflammaging alongside CRP.
Fibrosis
The formation of excess scar tissue inside organs. A normal part of wound healing when acute, but a destructive process when it occurs chronically and without injury. Fibrosis in the heart, liver, and kidneys progressively reduces organ function and is one of the primary ways IL-11 accelerates physical aging.
Hallmarks of aging
The Hallmarks of aging is basically the definitive list of everything that goes wrong in our cells as we get older. It first appeared in the journal Cell about a decade ago, and it completely changed the way we look at longevity. Instead of just seeing “aging” as one big mystery, this framework breaks it down into 12 specific cellular failures, things like chronic inflammation or cells losing the ability to recycle their own waste. This is exactly why the new data on IL-11 is so exciting. Blocking that one protein is a massive shortcut because it hits at least four of these hallmarks at the same time.
hsCRP (high-sensitivity CRP)
A more precise version of the standard CRP blood test. It detects very low levels of inflammation that a standard CRP test would miss. Below 1.0 mg/L is considered optimal. Above 3.0 mg/L suggests elevated chronic inflammation and increased cardiovascular risk.
Idiopathic pulmonary fibrosis (IPF)
IPF is a terrifying condition because the lungs basically turn to scar tissue for no apparent reason. It is the clearest proof we have that IL-11 is a major driver of human disease. Because that connection is so undeniable, scientists are using IPF as the main testing ground for new drugs that block this protein. If these trials work, it would be a massive win, not just for lung health, but for our understanding of how to stop organ damage as we age.
IL-6 (Interleukin-6)
IL-6 is an inflammatory protein that tends to increase as you age. For years, it was the usual suspect for everything from heart disease to diabetes. We specifically thought it was the main reason our organs get stiff and scarred as we get older. But the science has shifted. It turns out IL-6 is more of a supporting player, while IL-11 is the protein actually responsible for the damage. It is still a useful marker to track, but it is no longer the “main character” in the study of age-related fibrosis.
IL-11 (Interleukin-11)
IL-11 is the protein at the heart of this research. It is an inflammatory molecule that starts to pile up as you get older, and it is effectively a wrecking ball for your health. It causes your heart and kidneys to scar over while speeding up the muscle loss that most people start to notice after 40. It also shuts down the internal repair systems your cells need to stay healthy. When scientists blocked this protein in older mice, those mice lived 22 percent longer. That is a massive increase. In longevity science, we usually get excited about a 5 or 10 percent jump, so a 22 percent gain is a total game-changer.
Inflammaging
Claudio Franceschi coined the word “inflammaging” to describe the background noise of inflammation that builds up in your body over the years. Unlike the swelling you get from a cut, this version is invisible. It is sterile and low grade, meaning you do not feel it, but it is effectively the engine behind the health problems we usually blame on just “getting old.” It is the reason aging is so closely linked to heart issues and metabolic decline. Think of it like a slow, steady heat that eventually warps the machinery of your cells.
InsideTracker
A US-based blood testing and biological age platform. It analyses up to 43 biomarkers, produces a biological age estimate, and provides personalised recommendations based on which values are out of the optimal range. Tests can be ordered online with a home blood draw.
Lymphocyte percentage
The proportion of white blood cells that are lymphocytes, a key part of the immune system. The balance between different immune cell types shifts with age in ways that reflect immune decline. It’s one of the nine markers used in the PhenoAge calculation.
mTORC1 (mechanistic target of rapamycin complex 1)
A protein complex inside cells that acts as a growth regulator. When active, it drives cell growth and protein production. When overactive with age, it blocks cellular repair and accelerates decline. Suppressing mTORC1 is one of the most studied pathways in longevity research. IL-11 blocking, exercise, and caloric restriction all suppress it.
NHANES (National Health and Nutrition Examination Survey)
NHANES is a massive US study that tracks the health and nutrition of thousands of people across the country. It is essentially the primary source for almost everything we know about how Americans eat and age. Dr. Morgan Levine used this data to build the PhenoAge clock because it includes decades of actual physical exams and lab results rather than just surveys. If you see a major headline about US health trends, the data likely came from here.
Omega-3 and omega-6 fatty acids
Two families of polyunsaturated fats with opposing effects on inflammation. Omega-3 fats (found in oily fish, flaxseed, and walnuts) tend to reduce inflammation. Omega-6 fats (found in many seed oils and processed foods) tend to drive it when consumed in excess. The ratio between the two in the modern diet is a meaningful factor in chronic inflammation.
PhenoAge
A biological age clock developed by Dr. Morgan Levine using data from the NHANES study. It calculates biological age from nine standard blood markers and has been shown to predict mortality more accurately than chronological age. An online calculator is publicly available, and the required markers are obtainable from standard bloodwork.
PREDIMED trial
PREDIMED was the massive Spanish trial that basically turned the Mediterranean diet into a global health standard. It tracked thousands of people for years to see how the food they ate affected their hearts and their inflammation markers. Most of the rock-solid science we have on why olive oil and nuts are good for you comes directly from this study. It is the one trial everyone in the nutrition space points to when they want to show that diet actually moves the needle on long-term health.
Quercetin
Quercetin is a plant compound you get from things like onions and green tea. It is a big deal in the longevity space because it flips the AMPK switch in your cells, essentially telling your body to go into “repair mode.” We have seen some great results in labs and a few human studies, but I would not say the case is closed on its anti-aging benefits for people just yet. It is an interesting molecule to track, though it is likely more of a supporting player than a primary fix.
Resveratrol
Resveratrol is the chemical in red wine that sparked the “miracle pill” headlines about a decade ago. It got famous because a few mouse studies suggested it could stop aging, but the excitement has cooled off now that we have more human data. The main problem is that our bodies are incredibly bad at absorbing it. Even if it can trigger your cell repair signals, you likely are not getting enough into your system to actually matter. It is one of those supplements that works great in a lab but often fails to do much once you actually swallow the pill.
Sarcopenia
The age-related loss of muscle mass and strength. It typically accelerates after age 50 and is driven in part by chronic inflammation, including IL-11 activity. Sarcopenia is directly linked to falls, reduced mobility, and metabolic decline in older adults. Resistance training is the most evidence-backed intervention for slowing it.
TNF-alpha (tumor necrosis factor alpha)
TNF-alpha is basically an inflammatory alarm bell that stays “on” as you age. Like its cousins IL-6 and IL-11, it tends to creep up over the years and fuel the chronic inflammation that makes getting older so much harder on the body. If you are looking to lower it, you don’t need to guess. Omega-3s and Mediterranean-style eating are the two things with enough clinical evidence to actually move the needle.
TruDiagnostic
This US lab looks at the chemical tags on your DNA to calculate your biological age. Instead of just giving you a flat number, it breaks the data down by organ. It also uses the DunedinPACE clock, which acts like a speedometer for how fast you are aging right now. If you want the most data possible without joining a clinical trial, this is the test to get. They ship internationally as well.



