Eight hours of sleep means nothing if you’re skipping the one stage where your brain actually clears out Alzheimer’s-linked proteins.
Most people think of sleep as the brain powering down. The reality is closer to the opposite. While you’re unconscious, your brain runs one of the most sophisticated maintenance operations in the body: a fluid-based purge that flushes out the toxic proteins that build up during waking hours. Miss the right stage of sleep, and that cleaning doesn’t happen.
This isn’t a metaphor. It’s a physical process with a name, a mechanism, and a growing body of evidence linking its failure to Alzheimer’s disease.
The Hidden Plumbing System Inside Your Brain
The brain presents a logistical problem. It’s one of the most metabolically active organs in the body, constantly burning fuel and producing waste. Yet unlike other tissues, it has no conventional lymphatic drainage. For most of modern science, how the brain cleared its own debris remained largely unexplained.
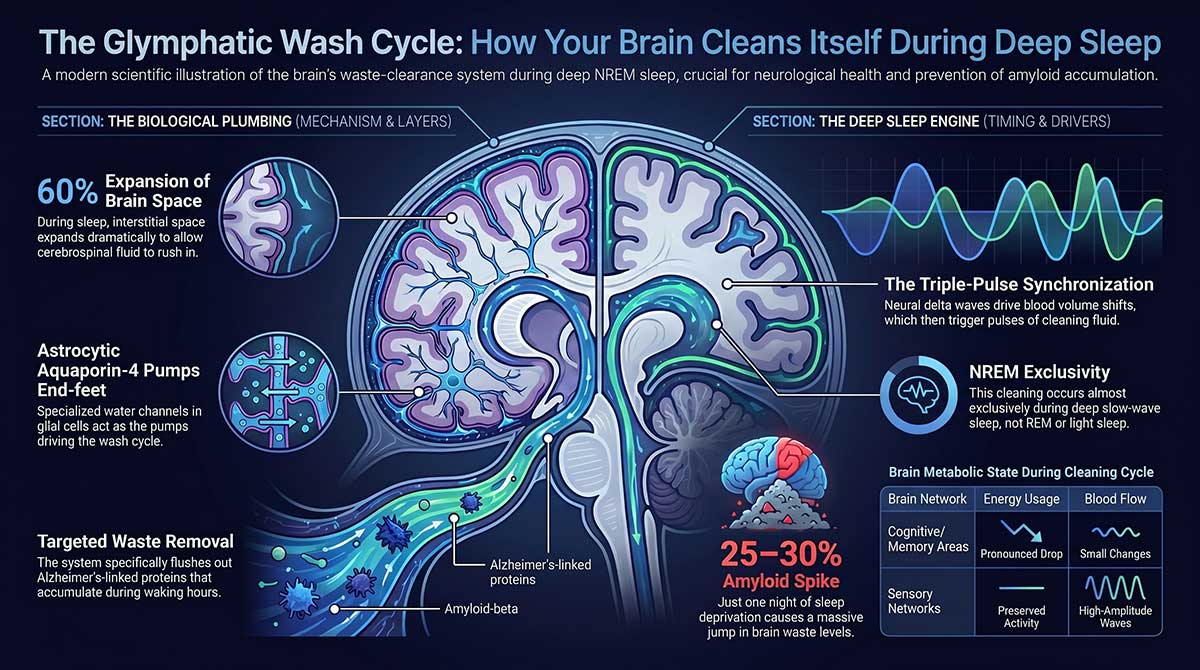
In 2013, neuroscientists at the University of Rochester published research that answered that question. Working with live mice, they discovered what is now called the glymphatic system, a brain-wide waste clearance network that operates through channels running alongside blood vessels. During sleep, cerebrospinal fluid (CSF) is pushed into brain tissue, mixes with the fluid surrounding cells, and carries waste products out. The name combines “glial” (referring to the support cells that drive it) and “lymphatic” (the body’s standard waste-removal system).
What made the finding so striking was the scale of the change. During sleep, the interstitial space (the gap between brain cells) expands by around 60%, allowing cerebrospinal fluid to rush in and dramatically increase the rate at which metabolites are flushed out. The brain physically opens up to let the wash cycle run. During wakefulness, that space contracts, and the system shuts down almost entirely.
The plumbing is driven by specialized glial cells called astrocytes. A 2012 study by Iliff et al. showed that cerebrospinal fluid enters brain tissue and exchanges with interstitial fluid through Aquaporin-4 water channels expressed in astrocytic end-feet that wrap around cerebral blood vessels. These cells act as the pumps. One of their primary targets is amyloid-beta, the protein that accumulates in the plaques characteristic of Alzheimer’s disease.
That connection between the glymphatic system and neurodegeneration isn’t incidental. It sits at the center of a rapidly developing area of research.
Why Deep NREM Sleep Is the Only Stage That Counts
People commonly associate sleep quality with REM sleep, the stage linked to dreams and memory processing. REM gets most of the popular attention. But for glymphatic clearance, the stage that matters is deep non-REM (NREM) sleep, sometimes called slow-wave sleep.
During deep NREM, the brain produces large, slow electrical waves known as delta oscillations. These aren’t just a sign of deep rest. They appear to actively drive the fluid dynamics that make cleaning possible.
In 2019, a research team led by Laura Lewis at Boston University used simultaneous fMRI and EEG to observe what happens inside sleeping human brains in real time. Fultz et al. found that during NREM sleep, large, slow electrical oscillations in the brain are coupled to oscillations in blood volume, which in turn drive waves of cerebrospinal fluid flow. The neural wave comes first, blood follows, and then a pulse of CSF sweeps through. The slow EEG waves that define non-REM sleep appear to be a key driver of the increased waste clearance that occurs during sleep.

This is the cleaning cycle in action. Without those deep, slow brain waves (the ones your body only produces during the NREM stage), the fluid pulses don’t happen at the same intensity, and clearance drops off. REM sleep, light sleep, and fragmented sleep don’t generate the same effect.
That distinction matters a great deal when you consider how modern sleep disruption tends to work. Alcohol, stress, blue light exposure, and age all disproportionately suppress deep NREM sleep, often while leaving total sleep time relatively intact. Someone sleeping seven hours but getting very little slow-wave sleep may be running significantly less cleaning than the numbers suggest.
What the Brain Is Actually Doing During This Phase
For years, scientists knew the glymphatic system was most active during NREM sleep but had limited visibility into the spatial details. A 2025 study from Massachusetts General Hospital began filling that gap.
Using a simultaneous tri-modal imaging approach that combined EEG, fMRI, and functional PET scanning, Chen et al. tracked how brain activity, blood flow, and glucose metabolism shift together as people fall into NREM sleep. The researchers identified two distinct patterns during NREM: sensory networks show high-amplitude, low-frequency blood flow waves with relatively preserved metabolic activity, while the areas associated with higher cognition and daydreaming show smaller blood flow changes and the most pronounced drop in energy use.

Your brain essentially splits into two operational modes during deep sleep. The parts that handle sensory input stay alert and keep burning fuel, maintaining a biological alarm system that can wake you if something demands it. The cognitive and memory areas, by contrast, power down as completely as possible. This helps explain how the brain stays responsive to the outside world even as conscious awareness fades.
That architecture isn’t a coincidence. The areas that shut down most completely during NREM are precisely the areas that benefit most from cleaning. The deep suppression of cognitive networks appears to be part of what allows the glymphatic system to run effectively, with reduced resistance and maximum fluid exchange.
The Cost of Skipping the Wash Cycle
When deep sleep is cut short, the cleaning cycle runs incompletely. Waste builds up. In the case of amyloid-beta, the accumulation can be rapid.
A 2018 study by Lucey, Bateman and colleagues at Washington University School of Medicine directly tested what happens to amyloid levels when sleep is disrupted. Using lumbar catheters to sample cerebrospinal fluid overnight, they found that sleep deprivation increased overnight amyloid-beta levels by 25 to 30% via increased production relative to sleeping controls. This happened in cognitively healthy adults. One disrupted night, and brain amyloid levels jump by more than a quarter.

That’s not a chronic effect of years of poor sleep. That’s the cost of a single bad night.
The effect is reversible. Amyloid levels come back down with recovery sleep, which suggests the system works as designed when sleep is sufficient. But for people with persistently poor sleep, or those who chronically sacrifice deep NREM for early alarms or late nights, the debt compounds over time.
Proof the Waste Actually Leaves the Brain
For years, a gap remained in the evidence. Researchers could show that amyloid levels in the brain rose with sleep loss. They could show that the glymphatic system was anatomically capable of clearing these proteins. But direct proof that sleep-driven glymphatic activity successfully moved amyloid and tau out of the human brain and into the bloodstream, where the body can eliminate them, was missing.
That gap closed in January 2026. A study by Dagum et al. at Applied Cognition published findings from a randomized crossover trial involving 39 participants. The researchers found that glymphatic clearance during normal sleep increased morning plasma levels of Alzheimer’s disease biomarkers compared to sleep deprivation. Higher levels of amyloid and tau in the blood after a night of normal sleep don’t indicate a worsening problem. They indicate successful overnight clearance: the proteins were moved out of brain tissue and into circulation for elimination.
Participants wore monitoring devices that tracked brain parenchymal resistance throughout the night. This is essentially a measure of how open or closed brain tissue is to fluid movement. The pattern matched what the glymphatic model predicts. When sleep was normal, resistance dropped, fluid moved freely, and morning blood samples showed elevated clearance markers.

It’s worth noting that the glymphatic field has active scientific debate around certain measurement methods. Some research groups have challenged specific aspects of earlier animal studies. But the human data, including the Fultz et al. 2019 imaging work and the Dagum et al. 2026 trial, provide increasingly strong direct evidence for the core mechanism in people.
Five Ways to Get More Deep NREM Sleep
Most sleep advice stops at “get eight hours.” That’s not wrong, but it misses the point. The goal is deep NREM sleep specifically. Total time in bed and time spent in slow-wave sleep are not the same thing.
Drop your core body temperature before bed. Deep NREM sleep requires a fall in core temperature. Taking a hot shower or bath 60 to 90 minutes before bed draws blood to the skin surface. When you step out, the rapid cooling that follows helps trigger the temperature drop your body needs to enter slow-wave sleep efficiently.
Stop eating at least three hours before sleep. Active digestion competes with sleep quality. The body can’t fully commit to restoration while processing a large meal. Cutting off heavy caloric intake well before bed removes one of the most common but underappreciated suppressors of slow-wave sleep.
Try pink noise. Research has shown that continuous pink noise (a slightly deeper, more balanced sound than white noise) can increase the amplitude of the slow brain waves produced during NREM sleep. Those waves, known as delta oscillations, run at 0.5 to 4 Hz, the slowest electrical rhythms the brain produces. This isn’t just about masking sound. The acoustic stimulation appears to synchronize and reinforce these exact rhythms, which are the same ones that drive the glymphatic pump.
Ventilate your bedroom properly. Carbon dioxide builds up in poorly ventilated rooms overnight. Even mild CO2 accumulation can trigger micro-arousals, brief partial wakenings that interrupt the continuous waves needed for effective glymphatic flushing. A slightly cooler, well-ventilated room supports deeper and more uninterrupted slow-wave sleep.
Get bright light within 30 minutes of waking. Morning sunlight anchors the circadian rhythm and determines the timing and strength of melatonin release at night. A strong, well-timed melatonin signal produces a faster and deeper dive into NREM sleep. This matters more than most people realise: deep NREM sleep is not evenly distributed across the night. It’s front-loaded. The longest and most efficient slow-wave cycles happen in the first half of the night. A well-anchored circadian clock ensures that NREM stage 3 arrives on schedule and runs fully. Skipping morning light pushes that window later and compresses it.
None of these require a supplement stack or an expensive gadget. They work by reducing the things that blunt deep sleep: poor temperature regulation, digestive load, arousal triggers, and circadian drift.
A word on alcohol. It’s the most common NREM disruptor that people don’t recognize as one. Alcohol is a sedative, so it feels like it helps you sleep. It does push you into unconsciousness faster. But it produces what researchers call alpha-delta sleep: a state where slow delta waves are constantly interrupted by faster alpha activity, the kind your brain produces when you’re quietly awake. The glymphatic pump requires sustained, uninterrupted delta oscillations. Alcohol effectively paralyses it while creating the subjective feeling of rest. You wake up having “slept” eight hours with very little of the cleaning done.
A Note on Where This Science Stands
The glymphatic field has moved fast and, in some areas, outrun rigorous human replication. Most foundational work was done in mice. The jump to human physiology is not always direct, and some researchers have raised methodological challenges to specific experimental approaches used in earlier animal studies.
That said, the human evidence has strengthened considerably. The Fultz et al. 2019 imaging study directly captured fluid-neural coupling in sleeping people. The Dagum et al. 2026 trial demonstrated successful overnight clearance of Alzheimer’s biomarkers in a controlled human crossover design. The Lucey and Bateman sleep deprivation study showed measurable amyloid accumulation in cognitively healthy adults after a single disrupted night. The Chen et al. 2025 imaging data mapped the regional brain dynamics of NREM with a level of detail not previously possible.
What remains less certain is the precise relationship between the nightly clearing measured in these studies and the decades-long trajectory toward dementia. Sleep deprivation research shows acute amyloid spikes. Epidemiological data links chronic poor sleep to elevated Alzheimer’s risk. Whether the two are directly causal, and what threshold of sleep disruption becomes dangerous over a lifetime, is still being worked out.
What is clear is this: the brain has a cleaning system, it runs at night, and it depends on a type of sleep most people don’t think about.



