Chronic stress doesn’t just exhaust you. Scientists found it physically changes a key brain region, and the recovery timeline will surprise you.
We all know chronic stress feels exhausting. The brain fog, the forgetfulness, the sense that your memory just isn’t working the way it used to. But can stress actually change the physical size of your brain?
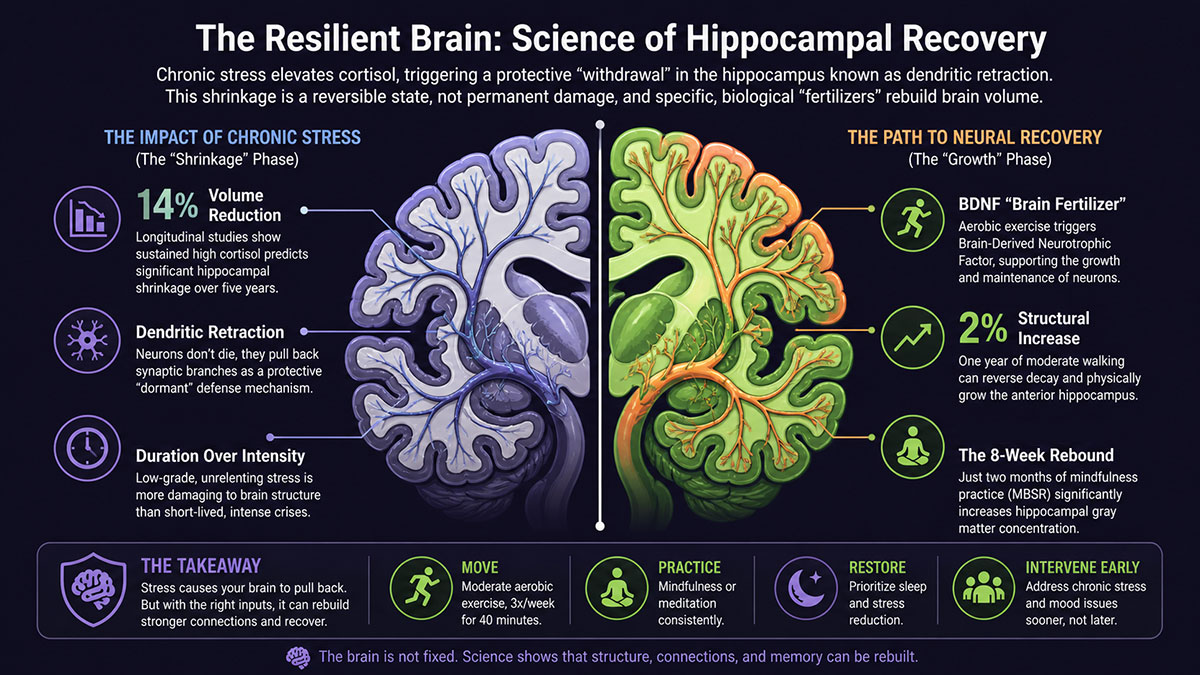
The answer, backed by decades of research, is yes. Long-term exposure to high cortisol acts like a slow drip on the brain’s memory center. Longitudinal studies tracking older adults over five years found that sustained high cortisol levels predicted a significantly smaller hippocampus, with some cases showing up to a 14 percent reduction in volume.
That’s a striking number. What most of those studies leave out is equally important: this process is not a permanent life sentence.
Modern neuroscience shows that the brain’s memory center is far more adaptable than we once thought. The damage stress causes follows a specific biological pattern, and that pattern can be reversed. Below are five research-backed truths about how your brain responds to chronic stress, and exactly what the science says you can do to rebuild it.
The “Shrinking” Is Actually a Defense Mechanism
When most people hear that stress shrinks the brain, they picture neurons dying off. That’s not quite what happens, at least not at first.
Neurobiologist Bruce McEwen’s landmark review published in Physiological Reviews (2007) documented what actually occurs at the cellular level under chronic stress. High cortisol concentrations make hippocampal neurons metabolically vulnerable. The cells don’t simply die. Instead, they pull back their synaptic branches in a process called dendritic retraction. Think of it like a tree dropping its leaves before a frost. The structure is still there. It’s just gone dormant.

McEwen’s review found that this synaptic withdrawal, particularly in the CA3 region of the hippocampus, represents a protective response to overstimulation. The structural architecture is paused, not permanently destroyed. His data showed that structural recovery can begin within weeks of the chronic stress ending.
This reframing matters a lot. The brain isn’t being damaged the same way a scar damages skin. It’s pulling back to protect itself, and it retains the ability to rebuild.
Primatologist Robert Sapolsky’s experimental work published in Science (1996) added an important layer to this picture. His research with primates found that high glucocorticoid levels make hippocampal neurons specifically vulnerable to injury, particularly in the presence of other stressors like reduced blood flow or excitotoxicity. His work established that neuroprotection is possible if glucocorticoid levels drop. The vulnerability, in other words, is conditional. Manage the cortisol, and the threat recedes.
Why Duration Matters More Than Intensity
People often assume that the worst stress is the most intense stress. A traumatic event. A sudden crisis. Research tells a more complicated story.
Neuroendocrinologist Sonia Lupien and her team at McGill University published a major longitudinal study in Nature Neuroscience (1998) tracking 51 healthy older adults over five years. They measured cortisol levels repeatedly across that period and used MRI and cognitive tests to assess brain structure and memory function.
What they found was clear: it wasn’t the biggest spikes of cortisol that predicted hippocampal shrinkage. It was the ones that never came down. Roughly one third of participants showed sustained high cortisol across the five-year window. That group performed significantly worse on spatial memory tasks and showed hippocampal volume reductions of up to 14 percent compared to participants whose cortisol levels stayed lower.
The takeaway: a single terrible month is far less dangerous than a year of low-grade, unrelenting stress. The drip wears down the stone.
Jens Pruessner’s theoretical work formalizing the “Glucocorticoid Vulnerability Hypothesis” (2010, published in Reviews in the Neurosciences) built on this foundation. Pruessner drew a specific distinction between neuron death and dendritic retraction, arguing that the retraction seen in chronic stress cases represents a specifically reversible form of plasticity. The structural architecture isn’t gone. It’s waiting for conditions to improve before it rebuilds.
What Untreated Depression Reveals About the Brain
Chronic stress and depression share a lot of biology. Both involve prolonged elevation of cortisol. Both show up in hippocampal volume loss. And research on untreated depression gives us some of the sharpest evidence for why early action matters.
Yvette Sheline and colleagues published an MRI study in The American Journal of Psychiatry (2003) examining hippocampal volume in women with a history of major depressive disorder. They found a specific relationship: the longer a depressive episode went untreated, the more predictable the hippocampal volume loss became. The number of days without treatment was a meaningful predictor of structural change, not just symptom severity.
This isn’t just relevant to people with clinical depression. It speaks to a broader truth about the brain under sustained cortisol load. Time spent in an unmanaged stress state adds up in a measurable, structural way. Sheline’s team also found that antidepressant treatment appeared to act as a shield against this volume loss, suggesting that intervention, whether medical, psychological or lifestyle-based, interrupts the process at the biological level.
The message is clarifying, not alarming. The sooner the stress system is addressed, the less structural work the brain has to undo.
Aerobic Exercise: Growing the Brain by 2 Percent
This is where the research gets genuinely exciting.
Neuroscientist Kirk Erickson and a team from the University of Pittsburgh ran a randomized controlled trial published in the Proceedings of the National Academy of Sciences (2011). They recruited 120 older adults and split them into two groups: one followed a supervised aerobic exercise program for a year, and the other did stretching. Both groups were assessed with MRI scans before and after.
The aerobic exercise group showed a 2 percent increase in the volume of the anterior hippocampus over the course of the year. The stretching control group showed a 1.4 percent decrease. That’s a roughly 3.4 percent divergence in brain volume between the two groups, produced purely by walking regularly.

Erickson’s team linked this growth to increases in brain-derived neurotrophic factor (BDNF), a protein that supports the growth and maintenance of neurons. Cardio exercise, it turns out, is one of the most reliable ways to increase BDNF levels in the hippocampus. Think of it as fertilizer for the brain’s memory center.
This study is often cited as proof of structural growth reversing stress and age-related decay. Subsequent meta-analyses have found mixed results when trying to replicate the exact magnitude of this effect across different populations. The underlying mechanism, aerobic exercise driving BDNF production and hippocampal growth, remains well-established. The 2 percent figure from Erickson’s trial represents one of the most cited single data points in the field, and it came from a well-designed, year-long controlled trial.
For practical purposes: you don’t need to run a marathon. The exercise protocol in Erickson’s study involved moderate-intensity walking, three times a week, building up to 40 minutes per session. That’s a genuinely achievable target for most adults.
→ See how blueberries support memory and brain function
The 8-Week Mindfulness Rebound
You don’t need a year-long commitment to see structural changes in your brain. Eight weeks may be enough.
Neuroscientist Britta Hölzel and colleagues published a study in Psychiatry Research: Neuroimaging (2011) examining what happens to brain structure after participation in a standard eight-week Mindfulness-Based Stress Reduction (MBSR) program. MBSR was developed by Dr. Jon Kabat-Zinn and combines meditation, body scanning and mindful movement into a structured group curriculum.
Hölzel’s team used MRI to measure gray matter concentration before and after the program in 16 participants, comparing them to a waiting-list control group of 17 individuals. The MBSR group showed measurable increases in gray matter concentration in the left hippocampus. No comparable changes appeared in the control group over the same period.
What makes this finding particularly significant is the timeline. Eight weeks. That’s two months of consistent practice, not years of intensive meditation. The brain responded to a structured reduction in stress perception with detectable structural changes in its primary memory region.
Changes were also observed in the posterior cingulate cortex, the temporoparietal junction and the cerebellum, regions linked to self-referential processing, emotional regulation and perspective-taking. The structural changes weren’t isolated to memory. The whole stress-response network appeared to shift.
Building a Lasting Neural Buffer
Recovery matters. Keeping it is a separate challenge.
A cross-sectional study by neuroscientist Sara Lazar and colleagues, published in NeuroReport (2005), compared long-term meditators with non-meditators. They found that people who had maintained a consistent meditation practice showed increased cortical thickness in regions tied to attention and the processing of sensory and emotional information, including the prefrontal cortex and the right anterior insula.

Cortical thickness in these areas is associated with better emotional regulation and a more measured, less reactive response to stress. Lazar’s team interpreted their findings as suggesting that long-term mental practice may build structural buffers against stress at the neurological level. The brain’s baseline response to cortisol spikes can, over time, be changed.
This doesn’t mean you need to become a lifelong meditator to protect your brain. But it does suggest that consistent stress management practice, whether that’s meditation, exercise, therapy or some combination, does something more than make you feel better in the moment. It may literally change the architecture of how your brain handles pressure.
That distinction is worth sitting with. For many people, stress management is the actual mechanism by which the brain repairs itself, not just a layer of comfort applied on top of damage.
What the Research Actually Tells Us
Chronic stress triggers a protective withdrawal response in the brain’s memory center. Cortisol, elevated for long enough, causes hippocampal neurons to pull back their branches and the overall volume of the region to decrease. This process is measurable on MRI and, in some cases, produces real cognitive consequences.
The brain is not passive in this process, though. The same structural plasticity that allows stress to cause shrinkage also allows recovery to produce growth. Aerobic exercise, mindfulness practice and early intervention in stress-related conditions all show measurable structural effects in the hippocampus. The research gives us approximate timelines: weeks for initial recovery when stress ends, two months for gray matter changes from mindfulness, one year of moderate exercise for a 2 percent volume increase.
Stress changes the brain in ways that, with the right inputs, can be changed back. That’s not false hope. That’s what the data says.